The understanding of molecular recognition processes requires a thorough examination from different perspectives including thermodynamic and kinetic aspects of the binding interaction and structural aspects of the interactants and the complex.
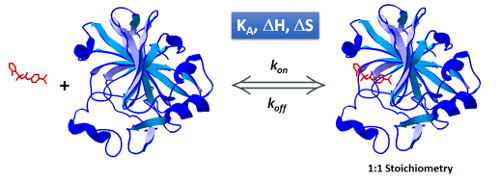
The determination of the binding affinity of two (or more) interactants (i.e. a protein/ligand system), through a steady state analysis provides information on how strong is the complex formed, and it is typically expressed in terms of equilibrium binding constants (association, KA , or dissociation, KD constants). The kinetic analysis of the interaction offers information on how fast the complex is formed and how fast it dissociates, expressed in terms of association and dissociation rate constants, respectively (kon and koff).
From steady-state and kinetic analysis one can get additional information (not just affinity and kinetic constants), i.e. the stoichiometry of the complex formed. Also, the experimental technique selected is a key determinant of the type of information that can be collected. For instance, Isothermal Titration Calorimetry (ITC) measures the heat released or absorbed during the binding event, and yet it provides a full thermodynamic characterization of the system including binding affinity (KA), enthalpy (ΔH) and entropy (ΔS). Moreover, a proper analysis of the ITC thermogram can yield kinetic information (1).
Nowadays, a complete understanding of particular biomolecular recognition processes is of fundamental importance and practical interest in the Pharmaceutical industry; i.e. The understanding of small ligand – biomacromolecule in the field of rational drug design (2).

In AFFINImeter we have implemented KinITC, this is a new method to obtain kinetic information from Isothermal Titration Calorimetry Data. With one single titration experiment it calculates the kinetic constants (kon and koff) and the thermodynamic data (KD and ΔH) of 1:1 binding interactions.
The new version is now available in AFFINImeter!
References
(1) Burnouf D1, Ennifar E, Guedich S, Puffer B, Hoffmann G, Bec G, Disdier F, Baltzinger M, Dumas P. kinITC: a new method for obtaining joint thermodynamic and kinetic data by isothermal titration calorimetry. J Am Chem Soc. 2012 Jan 11;134(1):559-65. doi: 10.1021/ja209057d. Epub 2011 Dec 16.
(2) a) Holdgate GA1, Thermodynamics of binding interactions in the rational drug design process. Expert Opin Drug Discov. 2007 Aug;2(8):1103-14. doi: 10.1517/17460441.2.8.1103; b) Murray JB1, Roughley SD, Matassova N, Brough PA, Off-rate screening (ORS) by surface plasmon resonance. An efficient method to kinetically sample hit to lead chemical space from unpurified reaction products J Med Chem. 2014 Apr 10;57(7):2845-50. doi: 10.1021/jm401848a. Epub 2014 Feb 24.