Title: Extending ITC to Kinetics with kinITC
Authors: Philippe Dumas, Eric Ennifar, Cyrielle Da Veiga, Guillaume Bec, William Palau, Carmelo Di Primo, Angel Piñeiro, Juan Sabín, Eva Muñoz, Javier Rial.
Abstract:
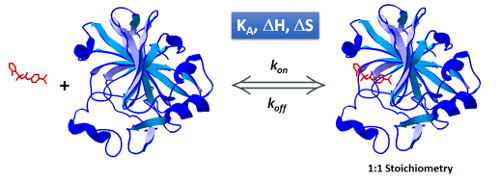
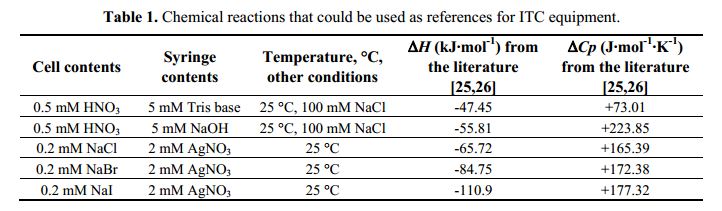
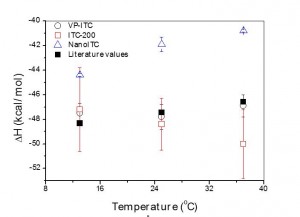
Isothermal titration calorimetry (ITC) has long been used for kinetic studies in chemistry, but this remained confined to enzymatic studies in the biological field. In fact, the biological community has long had the tendency of ignoring the kinetic possibilities of ITC considering it solely as a thermodynamic technique, whereas surface plasmon resonance is seen as the kinetic technique par excellence. However, the primary signal recorded by ITC is a heat power which is directly related to the kinetics of the reaction. Here, it is shown how this kinetic signal can be recovered by using kinITC, the kinetic extension of ITC. The theoretical basis of kinITC is detailed for the most common situation of a second-order reaction A + B Ω C characterized by kinetic parameters kon,koff. A simplified kinITC–ETC method based upon the determination of an “Equilibration Time Curve” (ETC) is presented. The ETC is obtained by automatic determination of the “effective end” of each injection. The method is illustrated with experimental results with a comparison to Surface Plasmon Resonance (SPR) data. kon values were obtained in a wide range, from 103 to 0.5 × 106 M− 1 s− 1. All procedures were implemented in the program AFFINImeter (https://www.affinimeter.com/).
Get full Publication here.