The Indian parable of “the six blind men and the elephant” tells the story of six blind men who touch an elephant in the hope of learning what it is like. As each one can only feel a different part of the animal the individual conclusions obtained are in disagreement and none of them provides a real view of the full elephant. “only by sharing what each of you knows can you possibly reach a true understanding”; that´s the moral behind this nice story.
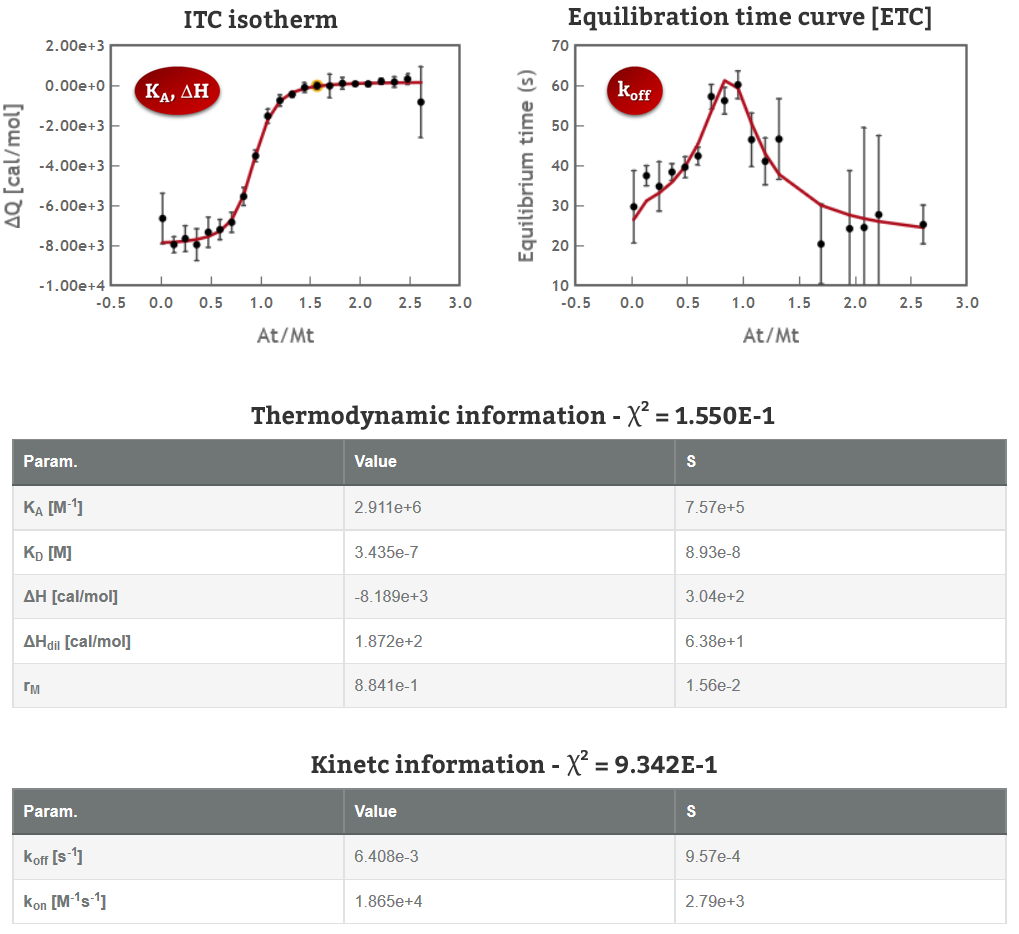
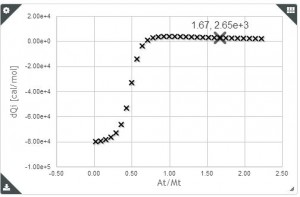
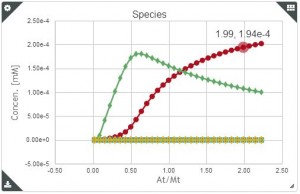
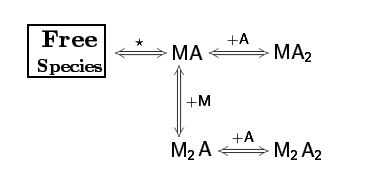
Sometimes, when we perform two or more ITC titration experiments of a complex interacting system under different experimental conditions (i.e. different concentrations and/or experimental setup) we find out that the individual analysis of the corresponding isotherms yields different values of the thermodynamic parameters. This result can be very confusing, especially for newcomers in the field of molecular recognition, because all these experiments are a representation of the same interaction and should converge to provide the same information. Frequently, the explanation for this behaviour is that each individual ITC experiment lacks of sufficient information to unequivocally determine the thermodynamic parameters of the binding event. “It´s like feeling only a separate part of the elephant”.
Analogous to the parable of the six men and the elephant, the way to get a true understanding of the binding event consist of the global analysis of the different isotherms.
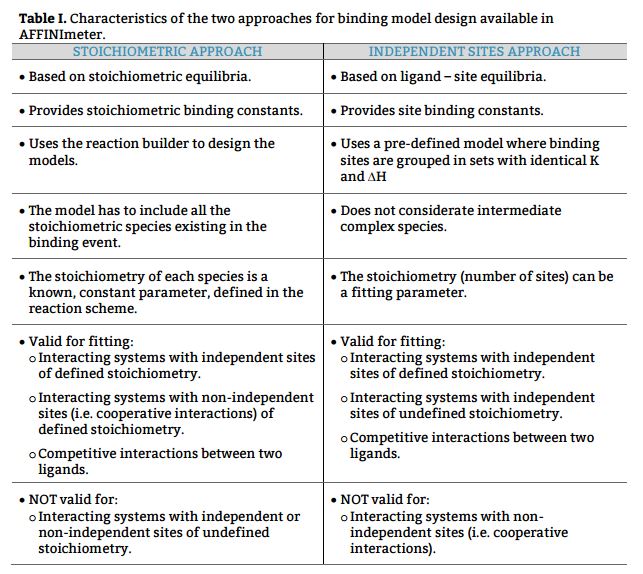
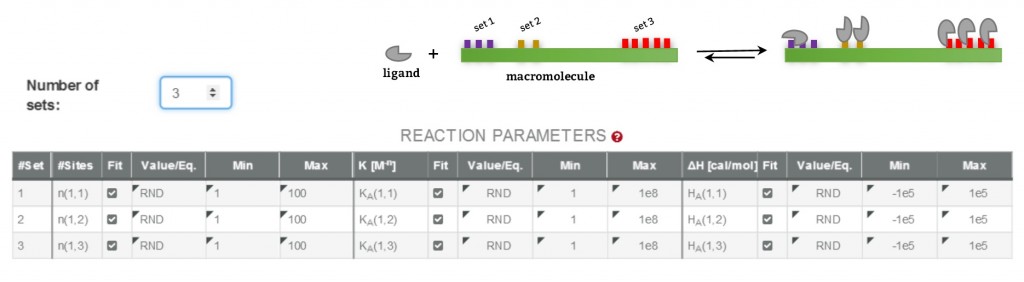
Being aware of the relevance of global analysis, in AFFINImeter we count with the possibility to perform Global fitting of multiple dataseries (isotherms) to tailored binding models where one or more fitting parameters are shared between isotherms. The number and identity of the parameters shared are selected by the user. Moreover, two or more parameters can be related trough mathematical relationships designed by the user. All these features make our global fitting tool the most potent among others to perform a robust analysis of ITC data of complex interactions.
Learn how to perform a global analysis with AFFINImeter: