Competition assays
Competitive binding assays where two (or more) ligands bind to the same receptor have become common experiments in many research areas, from basic investigations to innovation in the pharmaceutical industry. These assays can be done in different formats, i.e. through a displacement assay where ligand “L1” is displaced by ligand “L2” from a preformed complex “L1-receptor” or via titration of a receptor solution with a mixture of L1 +L2. Either way, the competitive binding assay provides rich thermodynamic and structural information of the various binding events taking place during the course of the experiment. Thus, Isothermal Titration Calorimetry (ITC) competition assays performed in a displacement format have been revealed as an efficient tool for the quantitative analysis of very high- / low- interactions, with application in the field of fragment based drug screening (ref).
Analysis of competition experiments with AFFINImeter
The versatility of the experimental setup in AFFINImeter-ITC permits the analysis of ITC competition experiments in its various formats. As an illustration, the following lines describe the analysis of an ITC isotherm resulting from a competitive experiment where a solution of a receptor in titrated with mixture of two competing ligands.
AFFINImeter contains a series of examples with which users can practice and learn the overall process of data fitting: from equipment and data uploading to fitting model design and data fitting.
In this post we will review an example of a competitive model fitting model used to analyze the experiment data of two ligands in the syringe competing for binding to the same receptor.
Practical case: competitive binding model
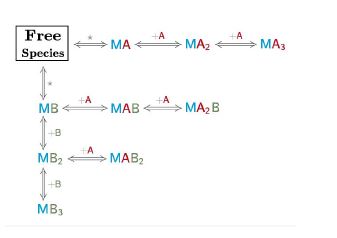
The AFFINImeter example “competitive binding model” illustrates an ITC experiment where two ligands, “A” and “B” compete with each other for binding to the receptor “M”.

This situation corresponds to a binding model consisting of three free species (A, B and M) two binding equilibria representing the interaction of M with A and M with B
The model was designed with the “reaction builder” and stored in “models”.The equipment used is decribed and stored in “equipments”. The dataseries is uploaded and stored in “dataseries”. When the dataseries is uploaded, the user has to complete the information relative to the equipment used and the species concentration. In this particular case (where there is a competitor “B”).
How to fit a project with AFFINImeter
1- Go to PROJECT MANAGEMENT and create a new PROJECT an a new FIT SUBPROJECT.
- Add the dataseries created previously to the subproject.
- Select the model created previously.
- Keep all the default values in FitSetting.
2- Press Run button.
This Steps are described in the following Video Tutorial:
You can follow this tutorial in AFFINImeter, the Experimental Data and Binding model are stored in your own AFFINImeter account. If you hasn’t registered yet go to the AFFINImeter Software WebPage to get your account.
Ref: W. B. Turnbull, Divided we fall? Studying low-affinity fragments of ligands by ITC. GE Healthcare Life Sciences protocol, 2011, pp 1-11.